The Future of Psychedelics As Medicinal TreatmentĬonforti believes that mounting data, along with FDA approval, will help push the envelope when it comes to wider acceptance of using psychedelics as medicine. The treatments are not self-administered, said Conforti, but are provided with guidance from trained therapists. Instead, a patient might have one therapeutic experience with MDMA and then another one six months later. “You’re not going to take an MDMA pill every day for the rest of your life,” explained Conforti. “If we have new therapies come to market, we’re going to be able to help more people.” Just like with new technology, Conforti said, there will be early adopters as well as those who come later to an understanding of the benefits of psychedelic medicine. “Novamind will continue to invest in bringing innovative mental health therapies to our clinics,” said Conforti. “We can create protocols, enroll patients in our groups, collect the data, and then offer evidence-based, innovative therapies at our clinics.” “With good reason - we’re talking about emerging therapies where, in some cases, none existed before.” Novamind hopes its clinical research will provide a benchmark for how administering these types of drugs will look after FDA approval.īecause of the combination of clinical sites and research programs, “We’re able to bring mental health innovations to life in real-time,” Conforti told Streetwise Reports. “There’s been a lot of excitement around the psychedelic sector,” said Conforti. Novamind also has three research sites in Utah that are co-located with the company’s clinics. “We’re building our business within the existing healthcare infrastructure,” said Conforti. Novamind also works with insurance companies, and many treatments offered at the company’s clinics are covered by insurance. In these clinics, psychedelic drugs are administered by licensed professionals. The company currently has seven outpatient mental health clinics and will soon be expanding to 10. “We had 35,000 visits to our clinics in 2021,” said Conforti. The company also offers curated programs such as a substance use disorder program and a program designed for frontline healthcare workers. Some of the unique programs offered by Novamind at its clinics include a palliative care program using ketamine in addition to psychotherapy to address high levels of anxiety and depression in patients with serious illnesses. Novamind’s CEO, Yaron Conforti, told Streetwise Reports that the company is specialized in “treating people who are suffering with mental health issues, including treatment-resistant conditions which the standard of care in pharmacology hasn't been able to treat.” The Novamind Approach to Treatment Is Unique The clinical trial is the first commercial study of LSD in 40 years and will investigate LSD’s ability to reduce anxiety symptoms in adults. (MNMD:NASDAQ), a clinical-stage biotech company focused on psychedelic drugs, is planning a Phase IIb trial that Novamind has been selected to host. The company also operates Cedar Clinical Research, a contract research organization that conducts clinical trials for psychedelic medicines including ketamine, psilocybin, LSD, and MDMA, and performs other evidence-based research on psychedelic medicine and how it can be used to enhance mental health. 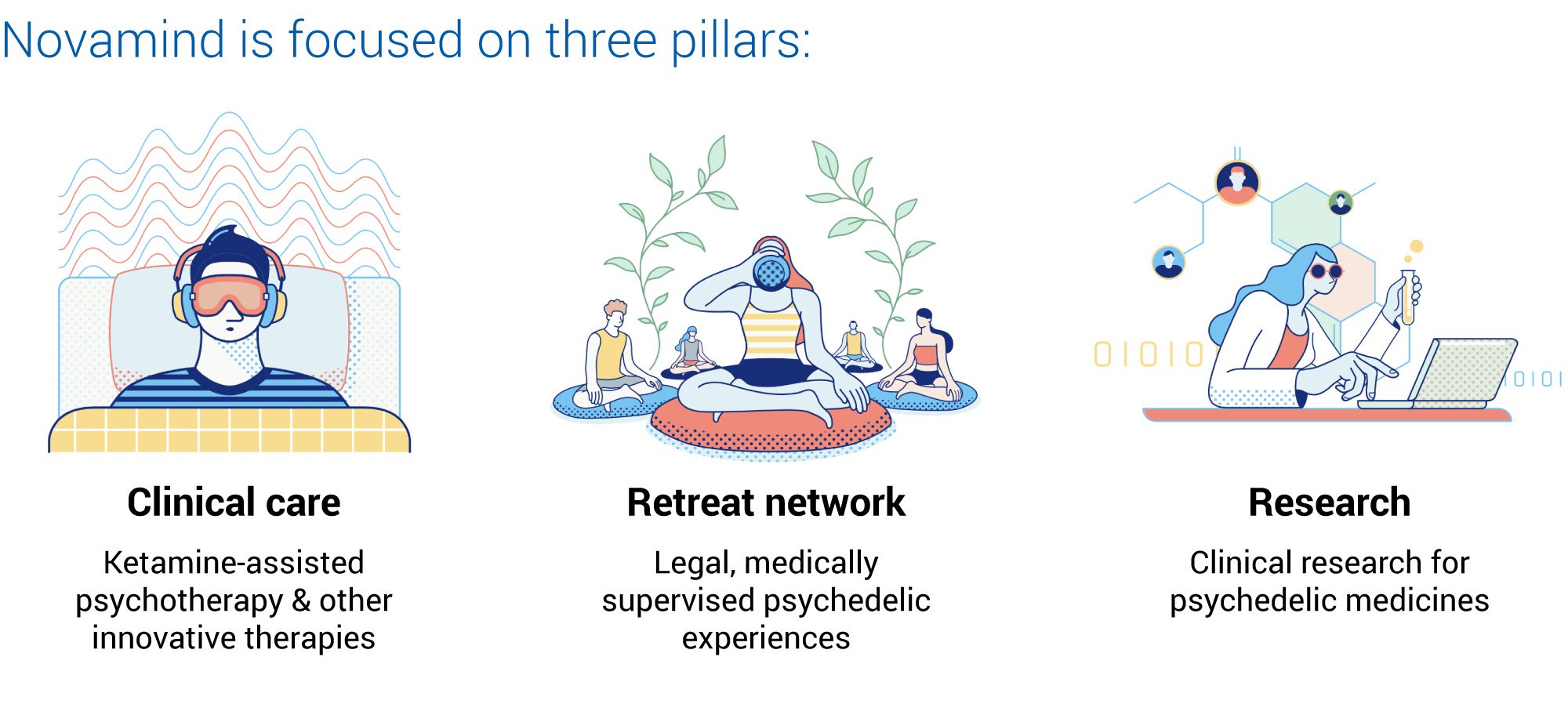
under the Cedar Psychiatry and Foundations for Change brands. Novamind enables safe access to psychedelic medicine through a network of integrative, outpatient mental health clinics operating in the U.S. Drug Enforcement Agency to store and administer psilocybin for research. In 2021, Novamind was awarded two Schedule 1 Licenses from the U.S. Today marks another exciting milestone for our team as we continue to support drug developers, not-for-profits, and academic partners to accelerate psychedelic research and improve patient outcomes." How & Where Psychedelics Can HelpĪn estimated 280 million people worldwide have depression, and psilocybin is being recognized as a potential new tool to address the condition. Paul Thielking, Chief Scientific Officer at Novamind, commented, "The Psilocybin Trial is a key step in moving psilocybin through the clinical development process and closer to regulatory approval. The market value of medical psychedelics is valued at $190 million and is expected to exceed $2.4 billion by 2026.ĭr. The trial will investigate the use of psilocybin - a naturally occurring psychedelic compound - to treat major depressive disorder (MDD), a move that the company hopes will nudge the dial toward FDA approval of the drug. (NM:CSE), whose mission is to increase access to psychedelic medicine for a range of mental health conditions, recently announced the company will be conducting a significant phase II clinical trial sponsored by the Usona Institute. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
